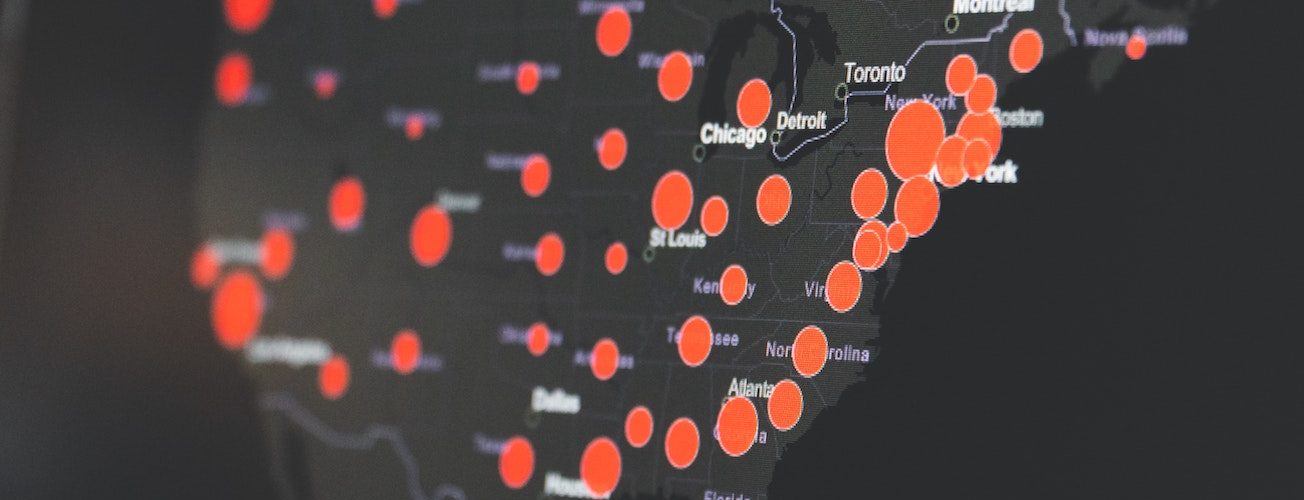
The Families First Coronavirus Response Act (FFCRA) went into effect in the U.S. on April 1, 2020, and it makes a lot of changes to sick leave for businesses and employees. It also has a significant impact on healthcare services, including provisions for medical services budget increases and full coverage for COVID-19 diagnostic testing and testing-related expenses through December 31, 2020.
Many organizations are concerned about the expansions to the Family and Medical Leave Act (FMLA), the Emergency Family and Medical Leave Expansion Act (EFMLEA), and the Emergency Paid Sick Leave Act (EPSLA). However, FFCRA includes exemptions for healthcare organizations, leaving many asking how the Families First Coronavirus Response Act affects healthcare services.
Just so you know
You’ve got your medical toolkit ready to help those in need — but what about your telemedicine toolkit? With Jotform’s HIPAA-friendly telehealth platform, you can easily create online medical forms that keep sensitive health data safe.
EFMLEA and EPSLA may not apply to healthcare workers
One of the provisions of the FFCRA expands the FMLA through the end of December. The expansion lets employees take leave if they are sick with COVID-19 symptoms or under a COVID-19 quarantine. During this leave, employers must pay them their regular wages for up to two weeks.
Additionally, this expansion allows employees to use this sick leave to take care of children in their care if the child’s daycare or school closed because of the pandemic. As with their own sick leave, their employers still have to pay them for up to two weeks, but only in an amount equal to two-thirds of their regular wages.
Employees may take leave longer than two weeks. They will receive payment of two-thirds their regular rate of pay (after the initial two weeks of leave) for up to 10 weeks.
However, healthcare workers may not be eligible for EFMLEA and EPSLA leave under some circumstances. The FFCRA specifically allows employers to deny leave to these employees.
The importance of proper coding
When it comes to billing and coding, healthcare service providers should note the requirements under the FFCRA. Providers will need to bill under the appropriate medical codes to ensure they’re reimbursed for services. They’re not allowed to collect fees, like copays, from patients for medical services related to COVID-19.
The FFCRA sets forth a set of COVID-19 CPT codes, which should make it fairly easy to calibrate billing, payment, and collection systems for testing services. The difficulty will be in making changes for doctor visits associated with testing; healthcare services will need to make sure these visits are flagged so that the organization can comply with billing requirements.
There is no guidance in the FFCRA about the pricing that insurance plans and healthcare providers must agree on for testing and testing-related visits. What’s important to note is that the Centers for Medicare and Medicaid Services (CMS) provided new codes to use for reimbursement related to COVID-19 testing and visits, which will probably set the standard for private reimbursement to providers.
Telehealth visit fees may be waived
Even before the COVID-19 outbreak, 76 percent of U.S. hospitals were using telehealth services. These services include video calls, remote monitoring, and phone consultations. Almost every state Medicaid program has some kind of provision to allow telehealth services, as do some private insurance providers. Medicare, however, offers only limited coverage for telehealth services.
Because of COVID-19, people are now using telehealth visits much more frequently. Some estimate that virtual doctor visits will double from 50,000 to 100,000 daily. Virtual visits can be used for triage in non-emergency situations, allowing patients to wait at home until lab testing is available rather than in a waiting room, as well as for routine visits like psychiatric medication adjustments.
The U.S. Department of Health and Human Services Office of the Inspector General is letting healthcare providers waive copays or pay less than the designated amount for telehealth visits as part of FFRCA, if the organization is seeking federal reimbursement. In addition, it’s possible that charging patients for telehealth visits will be forbidden in the future.
Registering for NDMS
If a healthcare organization might be providing COVID-19 testing to people without health insurance, they need to register with the National Disaster Medical System (NDMS). The NDMS has received $1 billion to reimburse healthcare providers for testing and services related to COVID-19 if they provide these services to uninsured people.
For healthcare services, FFCRA does pack a substantial impact. There is likely more to come as the law is put to the test in practice, so it’s important to pay attention to any new developments. Make sure that your billing, coding, and collection practices are compliant, and that you follow any EFMLEA provisions.




































































Send Comment: