Accurate, organized data is essential in clinical trials to develop new medical treatments. If there’s any doubt about the validity of the data, the entire trial can fail.
Collecting and maintaining clear data is important not only for good stewardship of money and resources, but it also has an impact on the lives of patients. Pharmaceutical providers have an ethical responsibility to bring safe, effective products to market.
There are a few key ways that you can improve data collection in clinical trials. Following these steps can help result in better data, cleaner forms, and a safer patient experience overall.
Use forms that automatically verify data
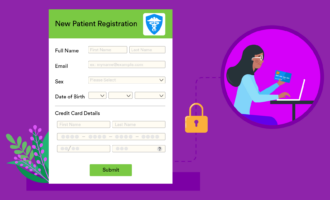
Intake forms are the foundation of your clinical trials. They provide a base level of information on patients, which you then build upon as you collect data. If you’re collecting the wrong data or using the wrong forms, then you risk a failed trial, which is why you need carefully developed case report forms (CRFs).
“Throughout the trial, certain clinical data is captured, as defined in the study protocol,” says Ayana Rowley Henderson, associate director of global regulatory affairs at Eli Lilly and Company. “Once the data is captured, it is sent via electronic gateway to the sponsor’s database. The sponsor then reviews the data for inconsistencies and may ask clarifying questions to the site.”
This review process used to be a manual one. However, the rise of eCRFs (electronic case report forms) has allowed trial developers to build alerts that catch incorrect data before someone submits it.
“A carefully crafted form will automatically warn users if they enter values that are not plausible, and will not let them submit forms until the error has been corrected,” says Tereza Benesova, senior customer success manager at Climedo Health, a platform for decentralized clinical trials. “In addition, data entry can be restricted to a specific range using drop-down menus or scales.”
For example, if a patient accidentally submits their weight in a field that requests height, the form will alert them and won’t allow the data to be submitted, as it’s outside a predetermined range. This prevents a 165-pound patient from accidentally reporting they’re 165 inches tall.
The more checks and reviews you can build into your intake forms, the cleaner your data collection will be in clinical trials. With automatic checks, trial developers can focus on more important, high-level tasks.
Develop inclusive best practices
One of the biggest challenges researchers face is finding participants for clinical trials. Many researchers need diverse samplings of people from a variety of backgrounds. If you only test your medication on one subset of the population, you might not realize how different people react to it.
In order to attract more participants — and assure them that they’re in good hands — develop inclusive intake forms. Even small changes in word choice can make future participants feel welcome.
The team at Queering Medicine created a useful guide for making your intake forms more inclusive. By making small adjustments to your forms, you can create a safer space for your LGBTQ+ trial participants. You can also help them better describe themselves, thus generating clearer data in your trials.
For example, there’s a significant difference between asking for a patient’s legal name vs the name they go by. Trans and nonbinary clients, for example, may have a different legal name in official records. The name they go by can also change over time, which means your trial may need to update these records on an ongoing basis.
Improve intake forms for decentralized trials
Decentralized trials take place outside of the clinic or research facility that developed them. These trials allow researchers to see how the product works outside of a medical setting, while reaching larger groups of people. Decentralized trials became essential during the COVID-19 pandemic when hospital visits were limited to emergency procedures.
One challenge of decentralized trials is managing the amount of data researchers receive. For example, if a patient uses wearable tech to track their heart rate and self-report symptoms, researchers can collect thousands of data points throughout the day, and they need to keep this data organized.
“The deluge of data not only needs to be managed, it also needs to be verified,” write Stacy Weil and Nicole Carswell at Premier Research. “After all, the patients responsible for much of the collection aren’t tech experts.”
As technology increases the number of data points available, data collection in clinical trials will require improved organization and data management.
Use Jotform for data collection in clinical trials
There are dozens of moving pieces involved in every clinical trial. You need a reliable system that allows you to quickly collect, review, and submit data.
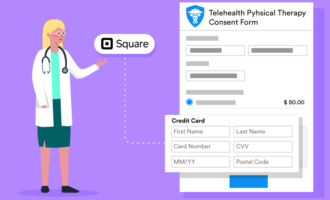
With Jotform’s easy-to-use online forms, you can collect all kinds of data and store it in a HIPAA-enabled manner. Jotform can make it easy for you to register new patients and track their progress in Jotform Tables. Learn how Jotform can improve your data collection in clinical trials and give our tools a try today.





























































































Send Comment:
1 Comment:
May 10, 2023
Jotform --
Clinical studies is a great use case for Jotform as I can attest. We are currently using Jotform in a fully decentralized pilot study for a Class II medical device. We are using Jotform to pre-screen potential participants; we use it for a daily study diary that participants complete, we use it to build eCase Report Forms for all of the online meetings between study participants and the clinical coordinators and to document and evaluate adverse events using Jotform workflow.
Our next step will be to conduct a pivotal study that would be submitted to the FDA for approval. However, we are concerned about using Jotform in the pivotal study because we can't find any information on whether Jotform is 21 CFF Part 11 compliant.
Is there somebody at Jotform we could speak to about your plans to validate and verify your platform to ensure it aligns with FDA regulations?
Best