Many clinical trials use a questionnaire to collect information from participants in a given study. This questionnaire is known as a case report form, or CRF, and it contains participant data so researchers can categorize and analyze it during the next phase of the research process.
Creating a proper CRF is an important step for researchers because it helps to standardize both the questions and the method used to collect data from each participant. This helps prevent biases or poor data collection from corrupting any part of the research. For the data to be valid, researchers must compare each participant’s responses directly with others’, making the standardization of CRFs paramount to a good study.
Traditionally, researchers wrote CRFs by hand or printed them on paper, but the increased use of digital technologies has meant that a lot of this data is now online. So, here’s a breakdown of why and how to create effective CRFs online.
What is a CRF in clinical trials?
Let’s dive a bit deeper into the purpose behind CRFs before explaining the need for electronic case report forms (eCRFs). When a patient is a participant in a clinical trial, researchers need to keep a record of all the effects and events of the trial. A patient’s CRF is exactly that, acting as a log for their participation, giving researchers a place to note any adverse effects that may have occurred during testing.
A CRF also contains all the questions and fields necessary to test the hypotheses under study, which means that the content of a CRF varies. One study of a certain kind of medication could include data on how long a patient slept after taking the medication, while another study, also using CRFs, could focus on a patient’s muscle mass before and after a physical therapy session.
There are no restrictions on the content in a CRF — it just needs to contain information valuable to the research at hand. Study sponsors are dedicated to making sure all the CRFs a clinical trial generates are accurate, even going so far as auditing that information after the study is complete. As a result, this data needs to be easily accessible and secure for the sponsor, researchers, and auditors alike.
Why should you move case reports online?
Like everything else in the modern world, CRFs are now shifting online. Unlike most things on the internet, though, the general public shouldn’t have access to CRF data. Clinical trials don’t typically release information about individual participants publically, so everything in an eCRF needs to be safe and secure.
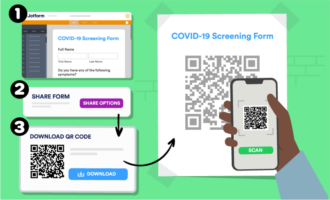
There are a number of advantages to using eCRFs as opposed to paper copies of CRFs. It’s faster and more efficient to fill out forms using computers, smartphones, and tablets. Just considering the amount of time it takes to write information by hand compared to typing is enough to prove that eCRFs can deliver huge productivity increases for researchers.
They’re also environmentally friendly compared to paper CRFs. Whereas the paper and supplies for physical CRFs usually travel great distances just to end up in a file cabinet, eCRFs are available online and are stored in the cloud.
ECRFs also ensure researchers can keep data indefinitely without needing a great deal of storage or spending time maintaining them. Clinical data from these records is also easier to share with other researchers, auditors, or study sponsors. Data in eCRFs is also more likely to be recorded accurately, without the need to interpret handwritten notes or the risk of data loss from damaged paper records.
How can Jotform help you create eCRFs?
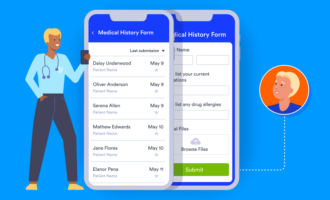
If you’re taking your research reports online, it’s a good idea to find a platform that can support all your needs before taking the plunge. It’s easy to create eCRFs with online form builder Jotform, which allows you to customize forms to your exact needs. If you’re not sure where to begin, Jotform already has hundreds of templates available to get you started quickly.
You can choose from templates for surveys (like ethnographic and correlational research surveys) and forms (like research evaluation, participant registration, research agreement, and consent forms). Whatever you need, there’s likely already a template available on Jotform that can help.
You can even allow researchers to collect their data through a smartphone or tablet with Jotform Mobile Forms, making data collection smooth and easy when conducting complex studies with a large number of participants. You can then collect and analyze it through the spreadsheet platform Jotform Tables, where you can sync responses collected from all forms for researchers to see as they go.
Jotform is also a highly secure platform, protecting sensitive data with features like a 256-bit SSL connection and form encryption capabilities. It also offers HIPPA-compliant forms and meets compliance requirements for regulations like the General Data Protection Regulation (GDPR).
Both healthcare organizations and pharmaceutical companies use CRFs to ensure their research is the best it can be. And, with Jotform, you can access all the benefits online forms can offer your study. As the world increasingly shifts toward online and virtual documentation, it’s a good idea to start learning how you can create the eCRFs you’ll need to keep up.
































































































Send Comment: